Info for Principal Investigators
Principles
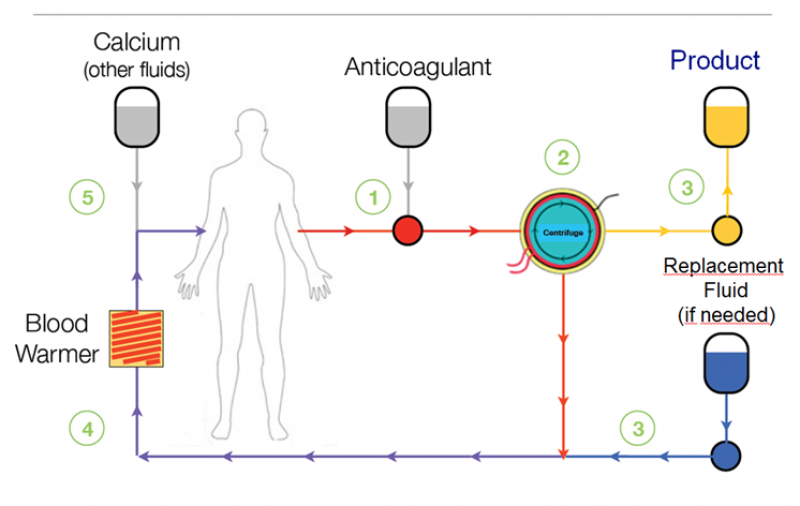
Leukapheresis is a well-tolerated, proven, safe, outpatient procedure which has been used in clinical practice for over 30 years. Leukapheresis is a blood collection technique during which leukocytes, including monocytes, are separated from the rest of the peripheral blood. The separation is performed at “Apheresis Centre” and the cells are separated in a device that is essentially designed as a continually working centrifuge (separator). Separated cells are collected into a certified blood bag which is then sent for further processing. Remaining blood components (erythrocytes, plasma and thrombocytes) are returned back into the patient’s bloodstream during the Leukapheresis.
LEUKAPHERESIS PROCESS
Apheresis Centres
SOTIO cooperates with Apheresis Centres which only use separators by well-established manufacturers. All cooperating centres are certified by related national regulatory institutions and regularly audited by SOTIO.
Separators
Apheresis centres use sophisticated devices (continually working separators) which adjust blood flow through the machine based on patient’s data. Separators are certified and regularly calibrated. During the collection sterile collection sets are used and disposed of after each procedure to minimize the risk of infection.
Leukapheresis Procedure
Leukapheresis is performed by trained staff at the Apheresis Centre. Prior to Leukapheresis the Apheresis Centre responsible person checks patient identity and provides additional information regarding Leukapheresis procedure if needed. If required by local regulations, patient is asked to read and sign an informed consent form. The Apheresis Centre staff then follows their standard procedures, usually involving measuring of patient temperature, blood pressure and pulse and doing a hematocrit test.
For the duration of the procedure patient is seated in a reclined position, with both arms resting comfortably on cushioned armrests. Sterile needles are placed into a vein in both arms and remain there throughout the procedure.
The Apheresis Centre staff (a trained nurse) then starts the Leukapheresis collection itself. In the most of the European countries a physician is present during the procedure and supervises it. Blood flows from the patient’s vein through the needle in one arm into the sterile apheresis separator and returns back to the patient’s body through the catheter in the other arm. Blood flow is adjusted according to blood pressure during the procedure.
Low dose of ACD-A anticoagulant (Anticoagulant Citrate Dextrose Solution) is used to prevent clotting as blood is pumped out. As dosing of anticoagulant citrate may cause hypocalcemia, oral or intravenous calcium supplements can be given if the patient experiences symptoms of numbness (symptom of low calcium level).
At the completion of the procedure, the needles are removed and the patient is examined to prevent bleeding and to check stable vital signs prior to discharge from facility.
The procedure usually takes 3-4 hours and the optimal volume of final product isapproximately 40-150 ml of leukocytes and fluid, depending on the separator type.
If necessary, a Central Line/Catheter via vena jugularis, via vena femoralis or via vena subclavia is considered. It is introduced either at the Apheresis Centre or arranged at contracted external healthcare facility.
Patient should be aware that Leukapheresis may not always be technically successful and may not succeed in obtaining the optimal volume of leukocytes (in 1-2 % of cases). The study drug is produced in an external laboratory during a complex procedure, therefore in case of failure to produce study drug, patient is informed about it approximately within four weeks after the Leukapheresis procedure.
Prior to Leukapheresis
Leukapheresis does not require significant changes in the patient’s routine or medication apart from the following:
- Patients are not allowed to use ACE inhibitors or angiotensin receptor blockers (sartans) 24 hours prior to Leukapheresis: depending on ACE-I / ARB administration time and scheduled time of Leukapheresis, patients might be required to skip multiple doses of their ACE-I / ARB medication so that a 24-hour window between last ACE-I / ARB dose and Leukapheresis start is always kept (no time limit is set for ACE-I/ARBs administration again once Leukapheresis is finished).
- Patients should stay well hydrated 2-3 days prior to Leukapheresis and avoid strenuous exercise, alcohol or caffeinated drinks 24 hours prior to the procedure.
- Healthy food rich in calcium is recommended; or calcium supplements may be considered by the Investigator.
Apheresis Centre evaluates the eligibility of the patient for the procedure prior to Leukapheresis. Patient’s general state of health, results of viral testing and suitability of vein access are the main evaluation points. The Apheresis Centre’s physician may also need to contact the Investigator and recommend changes in patient’s medication.
Post Leukapheresis
After the Leukapheresis the patient is adequately monitored for potential side effects.
Patient may experience increased physical weakness for a few days after Leukapheresis. Therefore it is advisable to avoid physical exertion. It is recommended to avoid lifting heavy loads as bleeding into the subcutaneous area at the needle insertion site may occur.
It is necessary to instruct the patient to rest, drink extra fluids and eat well.
